 Iron and steel rust when they come into contact with water and oxygen. They rust faster in salty water or acid rain. We call this process oxidation and aluminium oxidises also. Now here comes the confusing part. Aluminium corrodes but it does not rust. Rust refers only to iron and steel corrosion. When iron oxidise the color changes and it actually expands. This expanding and color change can produce large red flakes that we all know as rust. The expanding and flaking off of rust exposes new metal to further rusting. This is why it is so important to provide a barrier so rust doesn’t start.
Iron and steel rust when they come into contact with water and oxygen. They rust faster in salty water or acid rain. We call this process oxidation and aluminium oxidises also. Now here comes the confusing part. Aluminium corrodes but it does not rust. Rust refers only to iron and steel corrosion. When iron oxidise the color changes and it actually expands. This expanding and color change can produce large red flakes that we all know as rust. The expanding and flaking off of rust exposes new metal to further rusting. This is why it is so important to provide a barrier so rust doesn’t start.

Aluminium oxide corrosion also looks a lot more like aluminium or faded aluminium (dull gray to powdery white in color), so it isn’t as easy to notice as rusted iron. It is a good idea to coat the corrected aluminium with Ecocoat Rim to protect the metal from further oxidising and to lock in the mirror finish that you have perfected.
Aluminium oxidises almost immediately but this is a natural occurrence where the formation of aluminium oxide is able to protect the aluminium metal. This is because aluminium oxide is much more tough and resistant than the aluminium metal itself. The aluminium oxide is also less dense than the metal and so takes up more space than the metal and so as the metal is oxidised it puts the aluminium oxide in compression and this helps provide the surface with a good barrier layer that reduces the rate of any further corrosion. 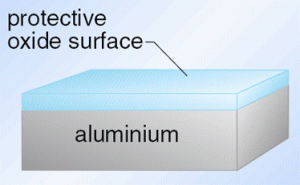
Due to this, the first monolayer of oxidation is virtually instantaneous solely depending on the arrival rate of oxygen. Beyond this the oxidation rate is dependent upon both the oxygen arrival rate and the rate of diffusion through the existing oxide layer. The average thickness of the oxide is around 1.5nm – 2nm almost immediately and reaching 2nm – 4.5nm within the period of 1 month to 1 year depending on the other variables. In most metallizers the residual gas within the vacuum system, even where the system can reach a good base pressure, means that the rate of arrival of oxygen to any surface is faster than 1 monolayer per second. Hence there is always plenty of oxygen available to oxidise the aluminium surface even in a vacuum system. It is also worth bearing in mind that even at the high aluminium deposition rates available in modern aluminium metallizers it is still common for there to be 1% – 2% oxygen contained within the coatings. Where oxygen is not available as a gas the aluminium is happy to strip the oxygen out of water molecules and as water is the dominant background gas of all vacuum systems there is no shortage of oxygen for oxidation to occur.
Aluminum Alloys
Wheels are normally cast from 5000 series aluminium alloy. This is based on the International Alloy Designation System which is the most widely accepted naming scheme for wrought alloys. Each alloy is given a four-digit number, where the first digit indicates the major alloying elements.
- 1000 series are essentially pure aluminium with a minimum 99% aluminium content by weight and can be work hardened.
- 2000 series are alloyed with copper, can be precipitation hardened to strengths comparable to steel. Formerly referred to as duralumin, they were once the most common aerospace alloys, but were susceptible to stress corrosion cracking and are increasingly replaced by 7000 series in new designs.
- 3000 series are alloyed with manganese, and can be work hardened.
- 4000 series are alloyed with silicon. They are also known as silumin.
- 5000 series are alloyed with magnesium.
- 6000 series are alloyed with magnesium and silicon. They are easy to machine, are weldable, and can be precipitation hardened, but not to the high strengths that 2000 and 7000 can reach. 6061 alloy is one of the most commonly used general-purpose aluminium alloys.
- 7000 series are alloyed with zinc, and can be precipitation hardened to the highest strengths of any aluminium alloy (tensile strength up to 700 MPa for the 7068 alloy).
- 8000 series are alloyed with other elements which are not covered by other series. Aluminium-lithium alloys are an example


